Mapping Platforms to Trials to Patents
Huggenberg from Stocktwits has provided all the data here and gets credit for this remarkable due diligence. An amazing amount of effort must have gone into mapping platorms to products to trials to results, and finally, patents.
(teachamantofish) am looking at options for how to create an interactive tool for this complex data.

This data is in progress (huggenberg continues to work), but I wanted to get this information out there so investors can position themselves pre-PDUFA with as much information as possible.
CAR-NK platform
We talked about CAR-T’s safety problems. Another drawback of CAR-T is its application in solid tumors, where CAR-T application remains challenging. Current CAR-T cell types remain incapable of treating solid tumors. And again, CAR-NK therapies from $IBRX are at the forefront. CAR-NK has several advantages and has the potential to close the therapy gap left by CAR-T cells in solid tumors. Last month, the first full report of a CAR-NK cell-based intervention in a solid tumor indication was published (HER2 t-haNK in Glioblastoma). Glioblastoma is the most common and aggressive primary brain tumor in adults. The conclusion is that CAR-NK in solid tumors is feasible and safe! IMO, CAR-NK will be the next big thing with the money inflow from the BP deal after Anktiva’s approval. There are exciting perspectives in CAR-NK, but there is still a long way to go. And yet another point to mention: CAR-NK can easily be combined with Anktiva or other cancer treatments. Compared with CAR-T cells, CAR-NK cells exhibit a greater variety of strategies to attack tumor cells. from: Link: https://jhoonline.biomedcentral.com/articles/10.1186/s13045-023-01492-8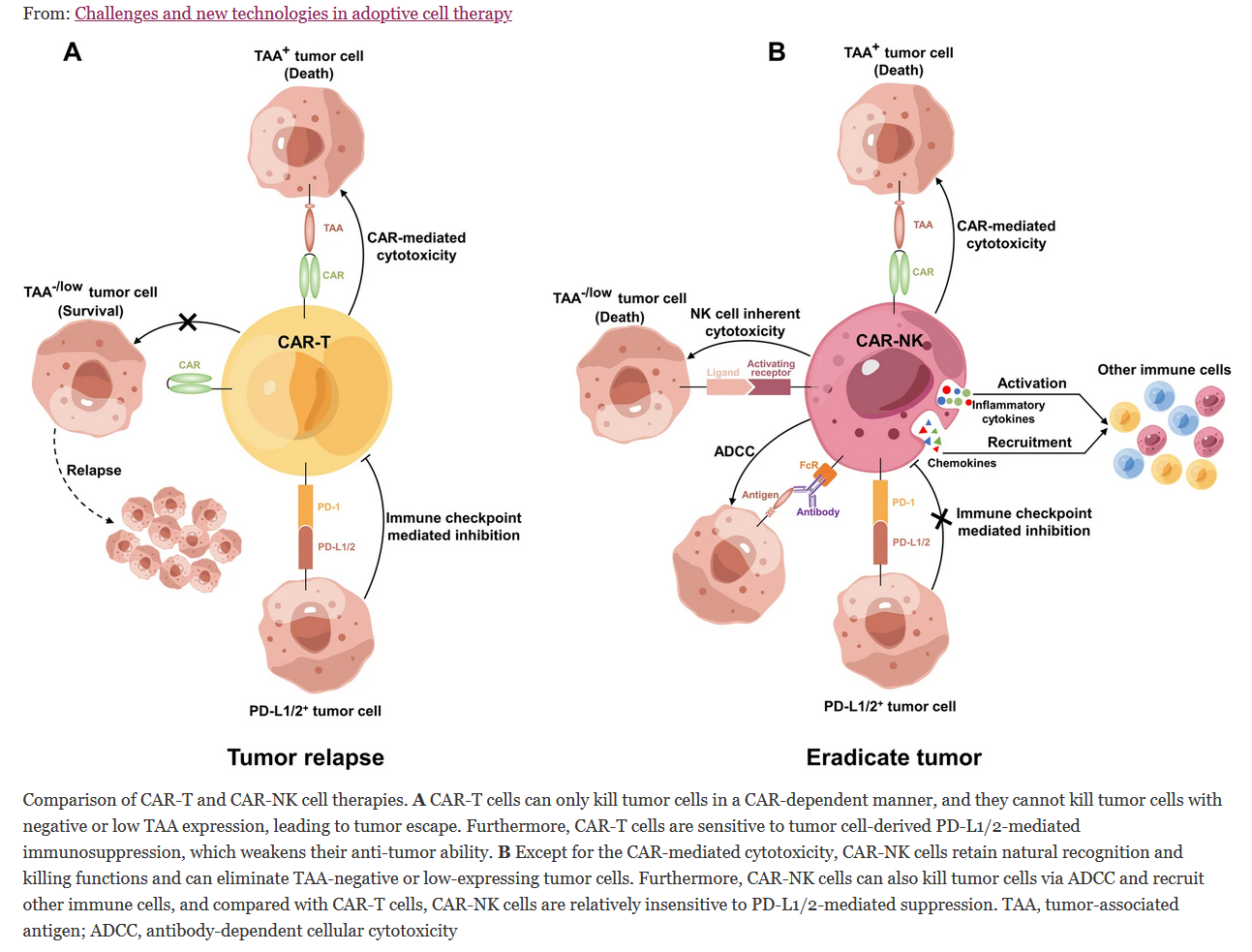
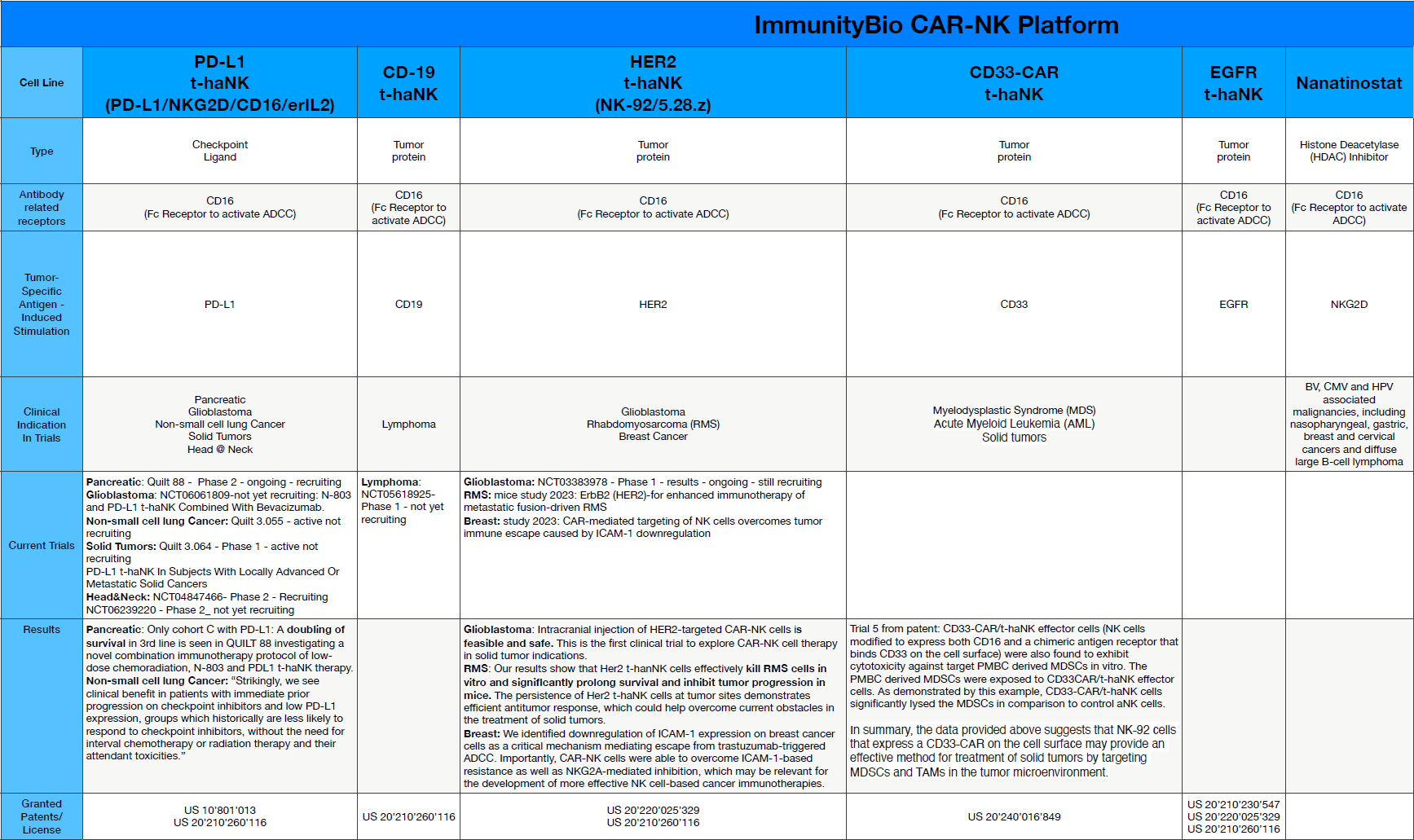
Future additions to the CAR-NK platform
Immunocytokine platform
What are ImmunityBio's future plans with Anktiva after approval? There are certainly efforts to approve Anktiva for other types of cancer, as many studies are already underway. The second way will be to expand the effect of Anktiva. A promising way is to add a second or third cytokine to Anktiva alongside IL-15. This would give us a bi- or tri-specific immunocytokine, which would multiply the range of applications of Anktiva in one fell swoop.Note that ImmunityBio is evolving N-803 into other drugs by combining it with cytokines with proven anti-cancer properties. For example:
- N-809 = N-803 combined with Anti-PD-L1
- N-809 = N-803 combined with CD20
- N-8xxx: N-803 is being tested in combination with IL21 (oHSV-21), IL-12, IL-18, and other cytokines.
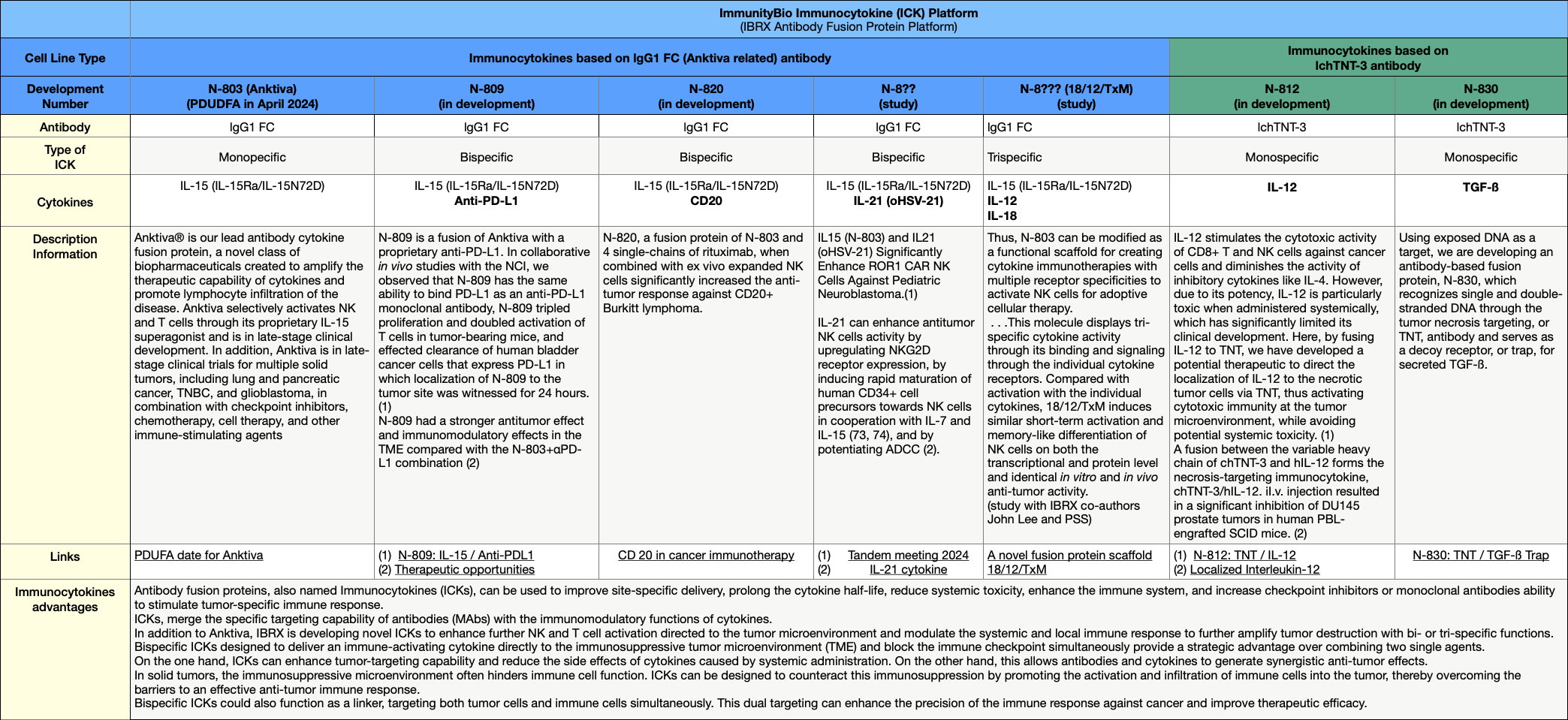
What is the value of this platform? In mid-2022, Roche acquired Good Therapeutics for $250M for a bispecific immunocytokine with the two cytokines PD1/IL2v. This application is not based on an approved base like Anktiva and has a long way to go. Immunitybio's platform can safely be estimated to be several times higher. https://medcitynews.com/2022/09/roche-shells-out-250m-to-acquire-good-therapeutics-and-its-preclinical-oncology-program/ The big point, IMO, is the BP collaboration to distribute Anktiva outside the USA. In addition to Anktiva, these new variants of Anktiva are certainly of great interest for BP, especially since one of these bispecific immunocytokines could improve the efficiency of one or more of their blockbusters (checkpoint inhibitors and/or monoclonal antibodies) and could also enable the route to solid tumors. Correction 01.26.2024: (N-8??: IL-15/IL21) deleted from the Excel sheet; drugs were not tested in the combo. IL21 is an external drug C021(an oncolytic viral herpes simplex virus). Preclinical study of Anktiva in Neuroblastoma, which most commonly affects children aged five or younger. „Conclusion: Our data demonstrated IL15 or IL21 based novel cytokine therapy (N-803 or C021) significantly enhanced the anti-tumor efficacy of ROR1 CAR NK targeting NB in vitro and in vivo.“ Anktiva and C021(an oncolytic viral herpes simplex virus) were tested separately. https://tandem.confex.com/tandem/2024/meetingapp.cgi/Paper/23519
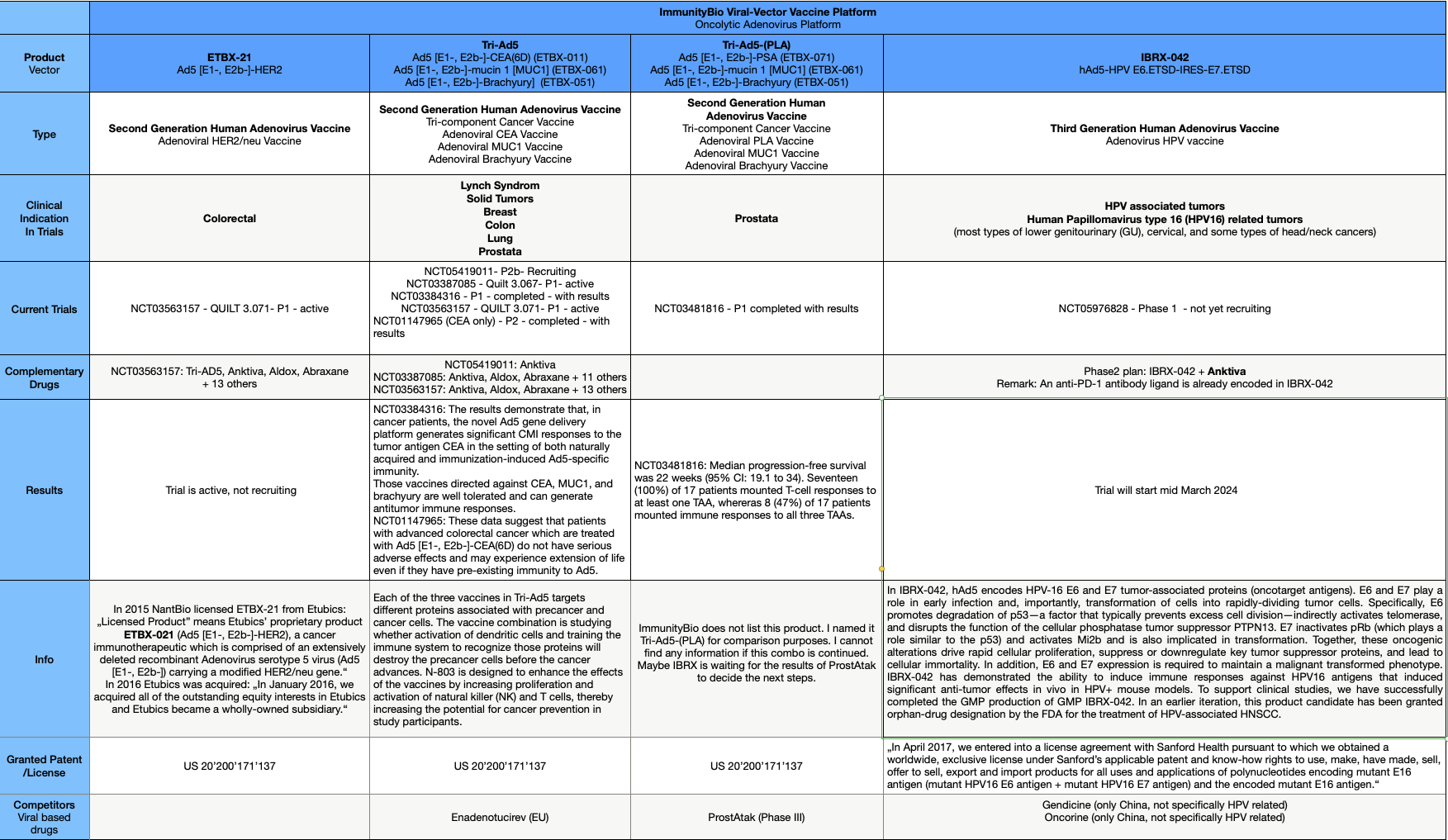
Viral vector vaccine platform
Details from the HPV trials from the $IBRX pipeline are now disclosed in clinical trials. The first step is the P1 trial, which examines the safety of IBRX-042. The second step will be combining IBRX-042 and Anktiva in a P2 trial.
HPV is the most common sexually transmitted infection. The Human Papillomavirus is known to cause approximately 95% of cervical and 30-60% of oropharyngeal carcinoma and many other cases. According to the Centers for Disease Control and Prevention, every year about 19,400 women and 12,100 men experience cancers caused by the human papillomavirus (HPV), which is the most common sexually transmitted infection in the U.S. Nearly all sexually active men and women are transmitted with HPV at some point in their lives. In most cases, HPV goes away on its own and does not cause any health problems. Some HPV infections, however, can persist for many years. Persistent infections with high-risk HPV types can lead to cell changes that may progress to cancer.
There are some prophylactic vaccines like Gardasil, but they are only effective if taken before exposure to the virus. But if you don’t get that vaccine, you still will have a minor chance of getting cancer. Researchers believe that it can take between 10 and 30 years from the time of an initial HPV infection until a tumor forms. There is no HPV-specific antiviral treatment for the infection. Until now, HPV-associated tumors are treated with surgery, radiotherapy, and chemotherapy or not HPV-related cancer and immunotherapy treatments.
But the two major oncotarget antigens E6 and E7 in IBRX-042 could change this. E6 and E7 are important components related to the Hallmark of cancer. E6 and E7 are major oncoproteins involved in cellular proliferation, invasion and metastasis, cell cycle arrest, angiogenesis, resisting cell death, tumor-promoting inflammation, genomic instability, evading growth suppressors, deregulation of cellular energetics, avoiding immune destruction and enabling replicative immortality.
IBRX-042 is the first oncolytic virus vaccine with E6 and E7 expression specifically related to HPV and will be a milestone if successful, as the E6 and E7 oncoproteins play a major role in many cancer pathways. Here is my overview of the IBRX oncolytic platform to show you the long way to IBRX-042.
https://www.mdpi.com/2076-0817/12/4/564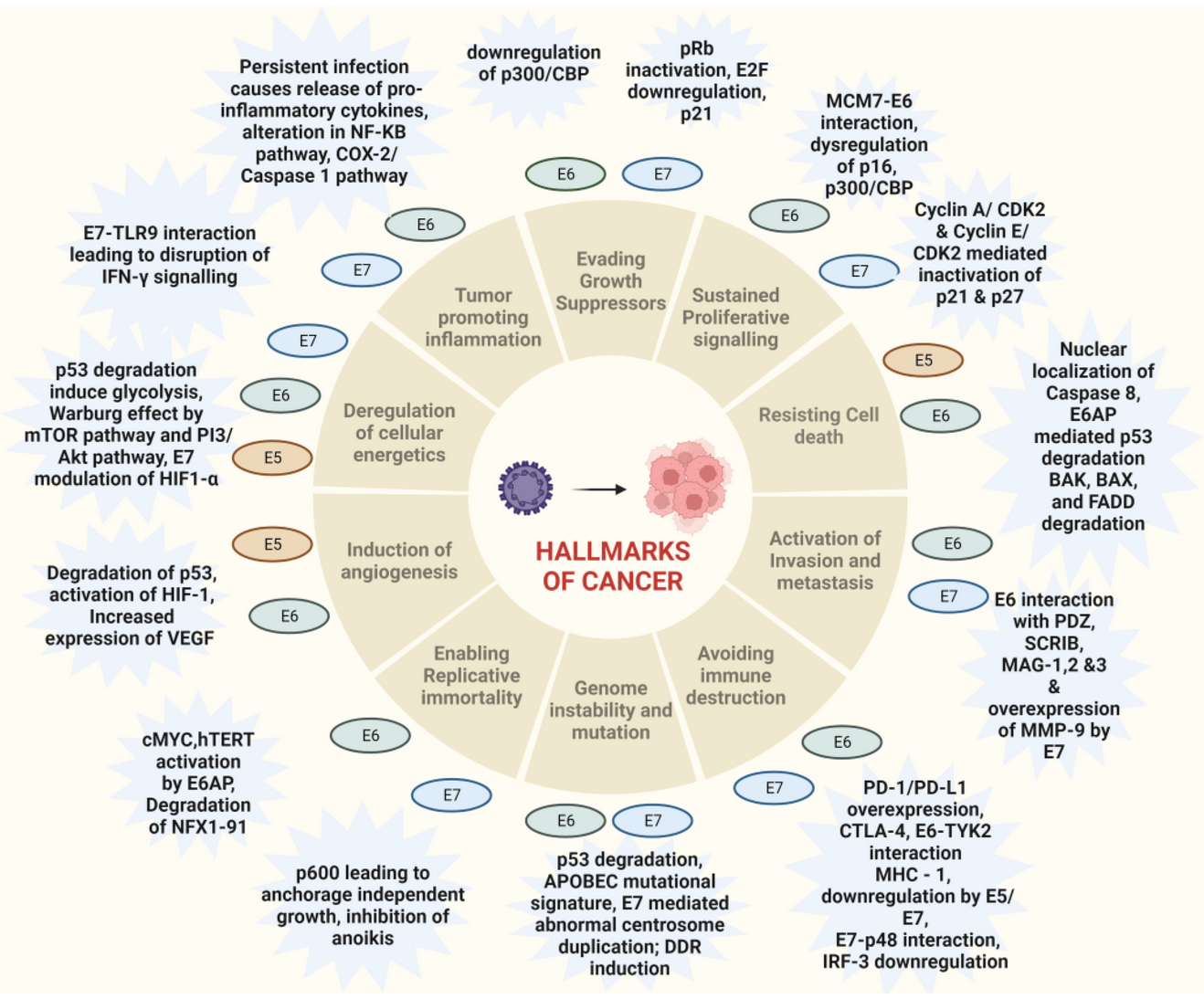
After Anktiva's approval, combining the oncolytic platform and Anktiva could be the next game-changer in cancer treatment, and the E6 / E7 oncolytic antigens could be used in a broader range of future applications. Further research
Covid notes
Covid may not be still pursued, but the technology behind sa-mRNA and SASA is superior to the vaccines from Pfizer and Moderna (see appendix). PSS developed this with the Advanced Health Institute AAHI, formerly IDRI, and PSS saved the institute from bankruptcy with $20 million and is now chairman of the board. https://www.aahi.org The technology can be used for future waves of infections and endemics and in IBRX future cancer vaccines.
